AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ionic solids2/20/2024 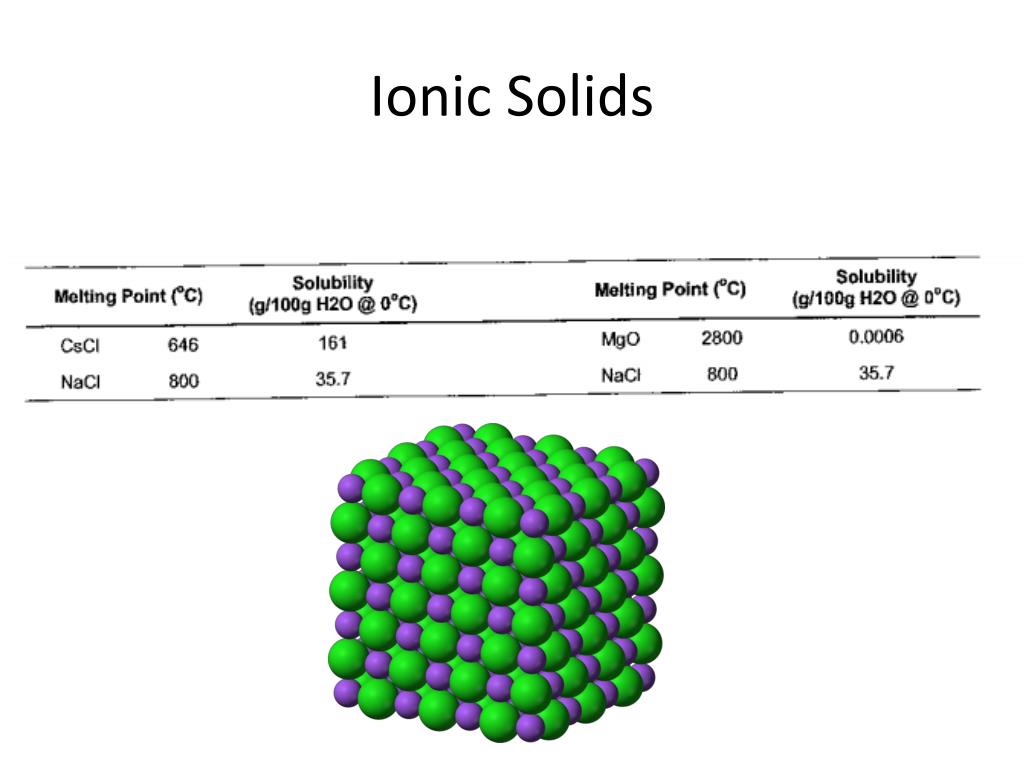
This Comment also clarifies how the charge of point defects fits with the standard. They have high density due to close packing of ions. Classical experiments from solid-state electrochemistry can be used to determine the charge of ions in solids. 2: Using Hess's Law to solve for an unknown enthalpy, the lattice enthalpy of formation for barium chloride. There are seven different lattice systems, some of which have more than one type of lattice, for a total of fourteen different unit cells, which have the shapes shown in Figure 12. 1: Description of Born Harbor energy cycle and its use to calculate the lattice enthalpy of formation for RbCl. A unit cell is defined by the lengths of its three axes (a, b, and c) and the angles (,, and ) between the axes. Ionic crystals are highly brittle because ionic solids contain parallel layers having cautions and anions in alternate positions. Bond Dissociation Energy (Cl-Cl) 244kJ/mol. Ionic crystals conduct electricity when they are in solution or in the molten states. Metals are generally malleable - that is, they can be hammered or pressed permanently out of shape. Ionic crystals do not conduct electricity in the solid state because on account of electrostatic force existing between them the cations and anions remain tightly held together with each other and hence occupy fixed positions. A metal (from Greek mtallon, 'mine, quarry, metal') is a material (an element, compound, or alloy) that is typically hard, opaque, shiny, and has good electrical and thermal conductivity. In the case of ionic crystals we always talk about the formula mass of these substances and not the molecular weight, because they do not exist in the form of molecules. ionic structure: permanganates, GROUP bonds accompanied alkali metal. Ionic bonds are formed from simple coulombic attractions of the ions opposite charges attract each other. For example NaCl and CsF have the same geometry because the radius ratio in both the cases is the same. The structure of the ionic crystals depends upon the radius ratio of cations and anions. The packing of the ions arrange themselves systematically in alternate manner. In Ionic solids their cations and anions attract each other and these forces are non-directional. Ionic bonds are formed between positive and negative ions because of their electrostatic attraction to one another. Ionic compounds without these ions are also known as salts and can be formed by acidbase reactions. Ionic compounds containing basic ions hydroxide (OH ) or oxide (O 2) are classified as bases. 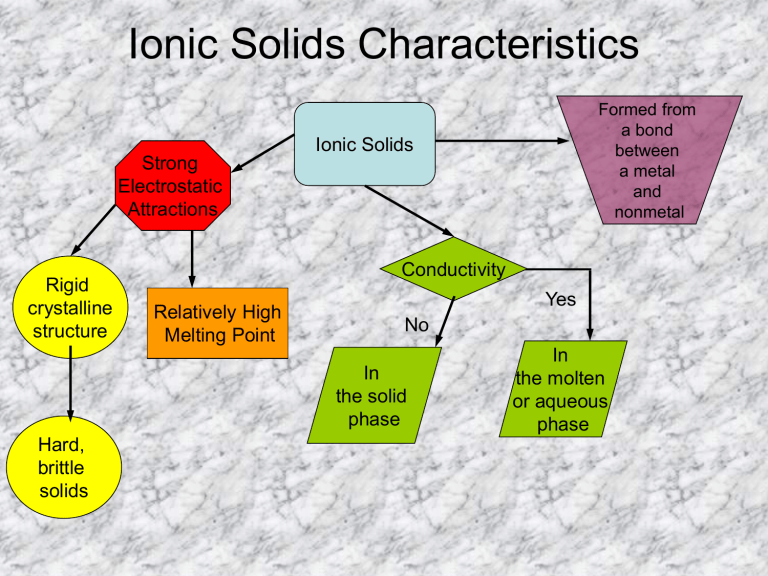
That is why ionic solids are very hard, have low volatility and high melting and boiling points. Ionic solids are a form of crystalline solid. Ionic compounds usually form crystalline structures when solid. Very high energy is required to separate the cations and anions from each other against the forces of attractions. Under ordinary conditions of temperature and pressure they never exist in the form of liquids or gases. So they are crystalline solids at room temperature. 
Properties of ionic Solids (1) Physical State:Ī well-defined geometrical pattern, describe the arrangements of the cations and anions.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
